4-(Hydroxymethyl)phenylboronic acid MIDA ester is an important organoboron compound widely used in modern organic synthesis, particularly in pharmaceutical research and advanced material development. As a member of the MIDA boronate family, it plays a crucial role in controlled cross-coupling reactions and stepwise molecular assembly. With increasing demand for efficient and selective synthetic building blocks, this compound has become a valuable intermediate in both academic and industrial laboratories.
The compound is commonly referenced in chemical databases and supplier catalogs under its full name 4-(Hydroxymethyl)phenylboronic acid MIDA ester, and is recognized for its stability, versatility, and compatibility with Suzuki–Miyaura coupling chemistry.
Chemical Identity and Structure
4-(Hydroxymethyl)phenylboronic acid MIDA ester is a boronic acid derivative in which the boron center is protected by N-methyliminodiacetic acid (MIDA). This protecting group significantly enhances the stability of boronic acids, which are otherwise prone to decomposition and undesired side reactions.
Key structural features include:
-
A phenyl aromatic ring
-
A boron atom integrated into a boronic ester framework
-
A hydroxymethyl (-CH₂OH) functional group at the para-position
-
A MIDA protecting ligand bonded to boron
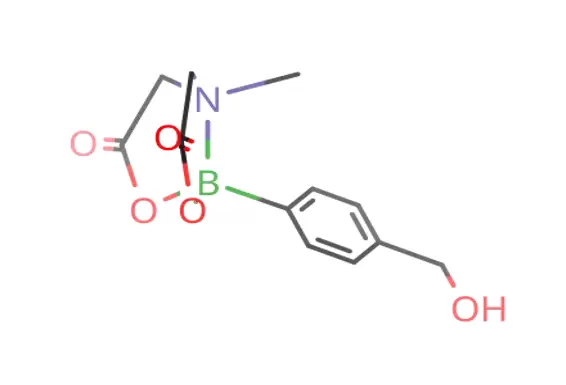
Molecular Information:
-
Chemical name: 4-(Hydroxymethyl)phenylboronic acid MIDA ester
-
Molecular formula: C₁₂H₁₄BNO₅
-
Molecular weight: ~263.05 g/mol
-
CAS Number: 1072960-82-7
The combination of aromatic functionality, hydroxymethyl substitution, and boronate protection makes this compound highly valuable in synthetic chemistry workflows.
Physical and Chemical Properties
One of the most important advantages of this compound is its improved stability compared to free boronic acids. The MIDA protection system reduces sensitivity toward moisture, air oxidation, and thermal decomposition.
Typical properties include:
-
Appearance: white to off-white crystalline powder
-
Stability: stable under dry and refrigerated conditions
-
Solubility: moderate solubility in polar organic solvents such as DMF and DMSO
-
Reactivity: controlled release of active boronic acid under mild hydrolysis conditions
The hydroxymethyl group provides additional chemical flexibility, allowing further functional transformation, such as oxidation to aldehydes or conversion into esters and ethers.
The Role of MIDA Protection in Boronic Acid Chemistry
The MIDA (N-methyliminodiacetic acid) protecting group is a key innovation in boron chemistry. It functions as a “slow-release” system for boronic acids, enabling precise control over reactivity.
Advantages of MIDA boronates include:
-
Enhanced Stability
Free boronic acids often degrade through oxidation or protodeboronation. MIDA protection prevents these issues. -
Purification Efficiency
MIDA boronates can be purified using standard chromatographic techniques without significant decomposition. -
Controlled Reactivity
The boronic acid is only activated under specific hydrolytic conditions, allowing stepwise synthesis. -
Compatibility with Iterative Synthesis
MIDA boronates are ideal for automated and modular synthesis approaches.
Because of these benefits, compounds like 4-(Hydroxymethyl)phenylboronic acid MIDA ester are widely used in modern synthetic design strategies.
Synthesis and Preparation Methods
The synthesis of MIDA boronate compounds typically involves the protection of boronic acids using MIDA ligands under controlled conditions.
A general synthetic route includes:
-
Formation of the corresponding phenylboronic acid precursor
-
Reaction with N-methyliminodiacetic acid in the presence of coupling agents
-
Controlled dehydration to form the stable MIDA ester
-
Purification through recrystallization or chromatography
The hydroxymethyl substituent is usually introduced before or after boronate formation, depending on the synthetic route and desired yield optimization.
Applications in Organic Synthesis
1. Suzuki–Miyaura Cross-Coupling Reactions
The most important application of 4-(Hydroxymethyl)phenylboronic acid MIDA ester is in Suzuki coupling reactions. These reactions are fundamental in forming carbon–carbon bonds between aromatic systems.
In this context, the compound acts as a masked boronic acid reagent, providing controlled reactivity and improving reaction efficiency.
2. Pharmaceutical Intermediate Synthesis
Many active pharmaceutical ingredients (APIs) rely on biaryl structures. This compound serves as a building block in the synthesis of:
-
Anticancer agents
-
Antiviral compounds
-
Anti-inflammatory molecules
-
Kinase inhibitors
Its stability ensures high reproducibility in multi-step drug synthesis pathways.
3. Chemical Biology and Probe Development
In chemical biology, boronic acid derivatives are used to construct molecular probes for studying biological systems. The hydroxymethyl group offers a convenient handle for conjugation with biomolecules such as peptides or fluorescent tags.
4. Iterative and Automated Synthesis
One of the most advanced applications is in iterative cross-coupling chemistry, where MIDA boronates are used in programmable synthesis sequences. This allows chemists to build complex molecules step-by-step with high precision.
Industrial Importance
The demand for MIDA boronate compounds has grown significantly due to:
-
Expansion of drug discovery programs
-
Increased use of modular synthesis techniques
-
Growth in agrochemical development
-
Rising need for efficient molecular libraries
4-(Hydroxymethyl)phenylboronic acid MIDA ester is particularly valuable because it combines structural versatility with operational stability, making it suitable for both small-scale laboratory research and industrial-scale synthesis.
Storage and Handling Guidelines
To maintain product quality and reactivity, proper handling is essential.
Recommended storage conditions:
-
Store in a cool, dry environment
-
Preferably at 2–8°C
-
Keep container tightly sealed
-
Avoid exposure to moisture and air
Safety precautions:
-
Avoid inhalation of dust
-
Use protective gloves and goggles
-
Handle in a well-ventilated laboratory
-
Follow standard chemical safety protocols
While not highly hazardous, it should still be treated as a laboratory-grade chemical reagent.
Advantages Over Conventional Boronic Acids
Compared with traditional phenylboronic acids, this compound offers several advantages:
-
Greater chemical stability
-
Reduced side reactions
-
Improved storage lifetime
-
Better compatibility with multi-step synthesis
-
Higher yield consistency in coupling reactions
These advantages explain why MIDA boronates are increasingly replacing free boronic acids in modern synthetic workflows.
Future Development and Research Trends
The use of MIDA-protected boronic acids is expected to expand further in the future, especially in:
-
Automated synthesis platforms
-
AI-driven drug discovery pipelines
-
Green chemistry applications
-
High-throughput molecular assembly
Researchers are also exploring new functional derivatives of hydroxymethyl-substituted MIDA boronates to expand their reactivity scope and biological applications.
Conclusion
4-(Hydroxymethyl)phenylboronic acid MIDA ester is a highly valuable organoboron compound that plays an essential role in modern synthetic chemistry. Its combination of MIDA protection and hydroxymethyl functionality makes it both stable and versatile, enabling its use in pharmaceutical synthesis, material science, and advanced organic reactions.
As chemical synthesis continues to move toward modular and automated approaches, compounds like this will remain critical building blocks for innovation in drug development and molecular engineering.
https://www.rlavie.com/industry/4-hydroxymethyl-phenylboronic-acid-mida-ester-structure-properties-a
rlavie
